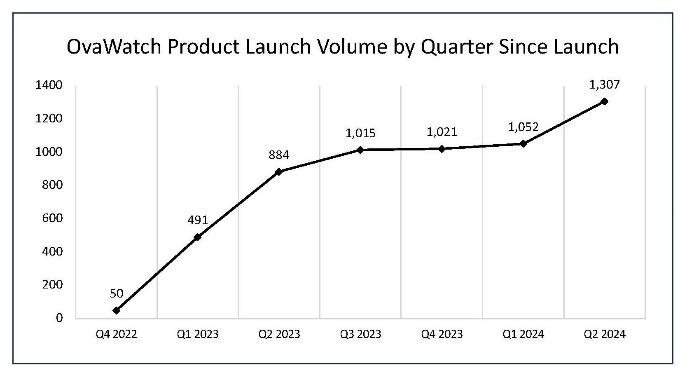
Preliminary OvaWatch® volume during Q2 2024 grew 24% over the prior quarter to 1,307 units
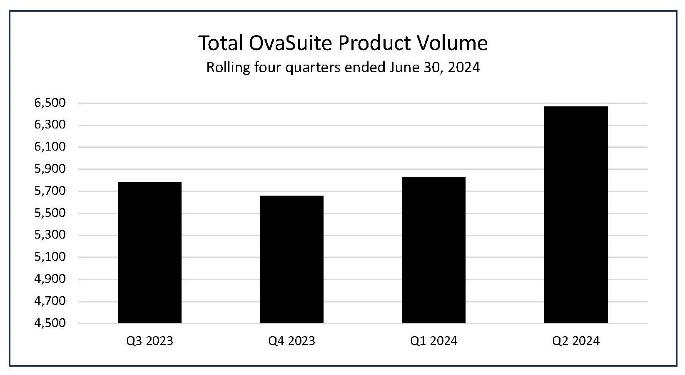
Preliminary OvaSuiteSM volume during Q2 2024 grew 11% over the prior quarter to 6,471 units
AUSTIN, Texas, July 10, 2024 (GLOBE NEWSWIRE) — Aspira Women’s Health Inc. (“Aspira” or the “Company”) (Nasdaq: AWH), a bio-analytical based women’s health company focused on the development of gynecologic disease diagnostic tools, today announced preliminary second quarter 2024 OvaSuite product volume and highlights.
Preliminary Second Quarter 2024 Product Volume and Highlights
- The number of OvaWatch tests performed during the second quarter ended June 30, 2024, was 1,307, an increase of 48%, compared to the 884 tests in the same period last year, and an increase of 24% to the 1,052 tests sequentially.
- The number of OvaSuite tests performed during the second quarter ended June 30, 2024, was 6,471, an increase of 3% compared to the 6,289 tests in the same period last year, and an increase of 11% to the 5,829 tests in the first quarter.
- The Company has completed a comprehensive analysis of its biobank and has identified up
to 70,000 serum, plasma, and whole blood samples that are available for secondary research. The Company is exploring opportunities to create non-dilutive sources of cash through related collaborations.
“Our OvaSuite product volume, and in particular our OvaWatch volume, has grown each month since the beginning of the year, demonstrating that the commercial changes we made last year are having a positive impact,” said Nicole Sandford, Chief Executive Officer of Aspira. “The data showing OvaWatch’s clinical effectiveness is resonating with physicians and payers alike. We believe that OvaWatch, which at this early stage already represents 20% of total volume, will be a key component of Aspira’s future growth. The release of the periodic monitoring feature in May radically increased the size of the test’s addressable market to between 2 and 4 million tests a year. Armed with a mature and effective sales force and strong clinical data, the momentum is clearly building.”
Ms. Sandford added, “We continue to be laser focused on cash management. Earlier this week, we announced the closing of an at-the-market private placement with gross proceeds of nearly $1.9 million with many of our largest shareholders and company insiders demonstrating their support of the Company. As mentioned in our June update, we anticipate providing a downward revision of our cash use for operations guidance at the time of our quarterly earnings report.”
Dr. Sandra Milligan, President of Aspira, said, “We continue to explore additional grants and other non-dilutive sources of funding including academic collaborations. We believe that our extensive biobank and available samples position us as an ideal strategic partner for others innovating in women’s health.”
“With respect to our in-development pipeline, we continue to progress in the design and development of our molecular tests,” Dr. Milligan added. “We are pleased with the performance of the digital droplet PCR platform, a crucial component of a commercially viable test, and plan to provide a more detailed update on our quarterly earnings call next month.”
In addition to these second-quarter highlights, the Company recently completed a $1.9 million private placement equity financing with broad participation from certain existing shareholders and company insiders. Net proceeds from the offering will support Aspira’s ongoing commercial activities as well as general corporate purposes and working capital.
About Aspira Women’s Health Inc.
Aspira Women’s Health Inc. is dedicated to the discovery, development, and commercialization of noninvasive, AI-powered tests to aid in the diagnosis of gynecologic diseases.
OvaWatch® and Ova1Plus® are offered to clinicians as OvaSuiteSM. Together, they provide the only comprehensive portfolio of blood tests to aid in the detection of ovarian cancer for the 1.2+ million American women diagnosed with an adnexal mass each year. OvaWatch provides a negative predictive value of 99% and is used to assess ovarian cancer risk for women where initial clinical assessment indicates the mass is indeterminate or benign, and thus surgery may be premature or unnecessary. Ova1Plus is a reflex process of two FDA-cleared tests, Ova1® and Overa®, to assess the risk of ovarian malignancy in women planned for surgery.
Our in-development test pipeline is designed to expand our ovarian cancer portfolio and addresses the tremendous need for noninvasive diagnostics for endometriosis, a debilitating disease that impacts millions of women worldwide. In ovarian cancer, our OvaMDxSM risk assessment is designed to combine microRNA and protein biomarkers with patient data to further enhance the sensitivity and specificity of our current tests. In endometriosis, EndoCheckSM is the first-ever noninvasive test designed to identify endometriomas, one of the most commonly occurring forms of endometriosis. The EndoMDxSM test is designed to combine microRNA and protein biomarkers with patient data to identify all endometriosis.
Forward-Looking Statements
This press release contains forward-looking statements, as defined in the Private Securities Litigation Reform Act of 1995. Forward-looking statements involve a number of risks and uncertainties. Such forward-looking statements include statements regarding, among other things, the timing and completion of any products in the pipeline development and other statements that are predictive in nature. Actual results could differ materially from those discussed due to known and unknown risks, uncertainties, and other factors. These forward-looking statements generally can be identified by the use of words such as “designed to,” “expect,” “plan,” “anticipate,” “could,” “may,” “intend,” “will,” “continue,” “future,” other words of similar meaning and the use of future dates. Forward-looking statements in this press release and other factors that may cause such differences include the satisfaction of customary closing conditions related to the offering and the expected timing of the closing of the offering. These and additional risks and uncertainties are described more fully in the company’s filings with the SEC, including those factors identified as “Risk Factors” in our most recent Annual Report on Form 10-K, for the fiscal year ended December 31, 2023, and subsequent Quarterly Reports on Form 10-Q. If any of these risks materialize or our assumptions prove incorrect, actual results could differ materially from the results implied by these forward-looking statements. There may be additional risks that Aspira presently does not know, or that Aspira currently believes are immaterial, that could also cause actual results to differ from those contained in the forward-looking statements. In addition, forward-looking statements reflect Aspira’s expectations, plans, or forecasts of future events and views as of the date of this press release. Subsequent events and developments may cause the Company’s assessments to change. However, while Aspira may elect to update these forward-looking statements at some point in the future, Aspira expressly disclaims any obligation to do so, except as required by law. These forward-looking statements should not be relied upon as representing Aspira’s assessments of any date after the date of this press release. Accordingly, undue reliance should not be placed upon the forward-looking statements.
Investor Relations Contact:
Nicole Sandford
Chief Executive Officer
